Trg Osvobodilne fronte 13
1000 Ljubljana
e-mail: info@enfist.si
telephone: +386 40 324 828

Practical strategy for the NMR structural characterization and quantitative analysis of small organic molecules nowadays includes several complementary stages. First, structural connectivities are determined from two-dimensional through-bond correlation experiments. Next, the relative stereochemistry is deduced from NOE correlations and coupling constants. Finally, the proposed structure is verified by a careful inspection of all of the observed NMR data. Modern NMR infrastructure allows analysis of samples of low concentrations, overlapping peaks and even high molecular weights. Once chemical structure is confirmed, determining the quantity of sample of a pure material, or as part of a mixture, is both simple and accurate.
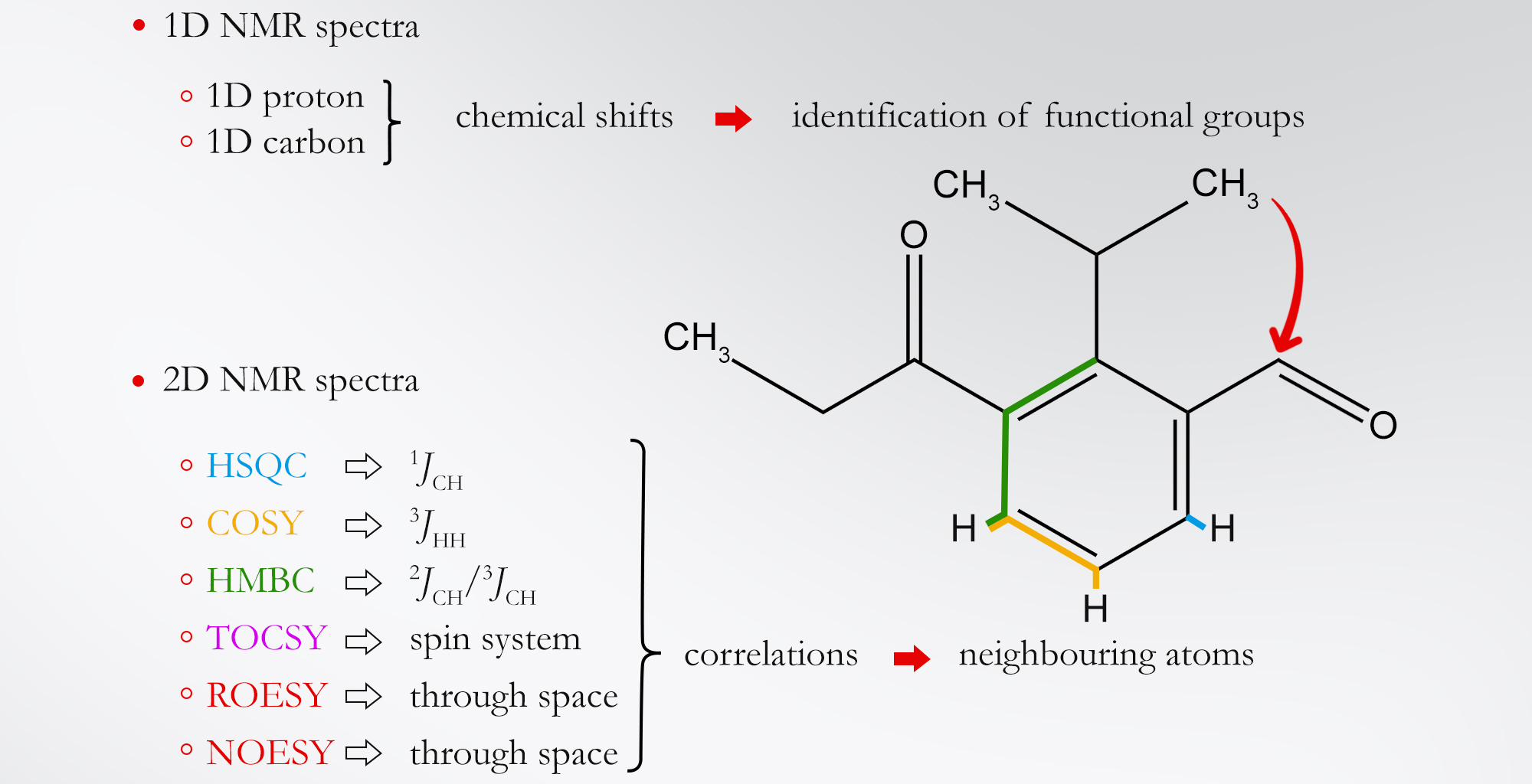
1H and 13C NMR spectra provide information about atoms included in the structure in terms of their electronic surroundings, their proximity to each other both in terms of scaffold connections and through-space interactions. Furthermore, 2D NMR spectra contain information about both through-bond and through-space interactions between atoms and are the most informative, especially when multiple types of 2D NMR spectra are obtained and analyzed in parallel.
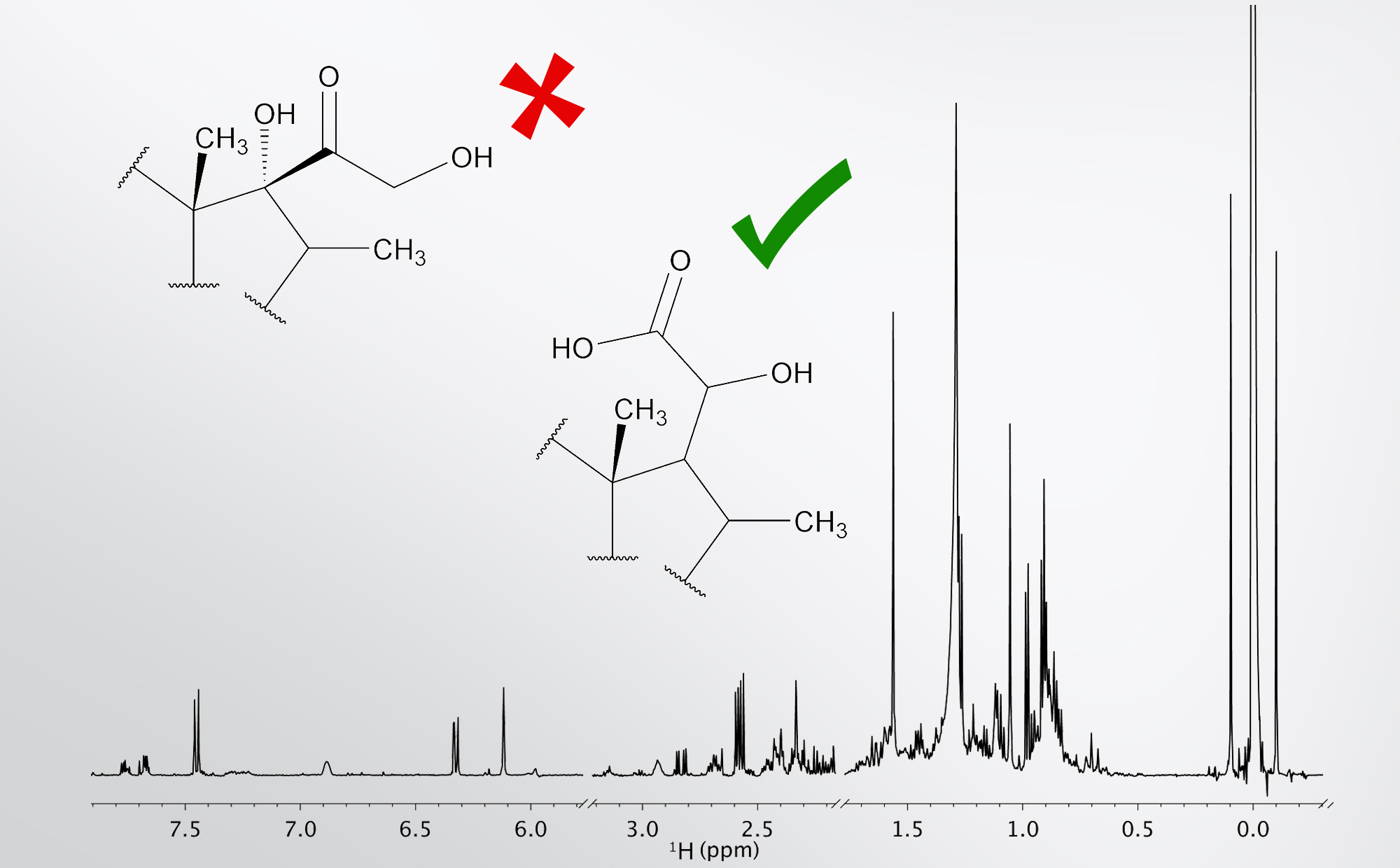
Combination of 1D and 2D NMR spectra allowed elucidation of chemical structure of minor component in complex mixture. We confirmed 2-hydroxy acetic acid moiety in the analysed substance as shown in the figure above. Afterwards, we determined its quantity, scaled to a reference sample, which was 60 μg.
Trg Osvobodilne fronte 13
1000 Ljubljana
e-mail: info@enfist.si
telephone: +386 40 324 828
EN-FIST Centre of Exellence, Copyrights reserved