Telephone: 01/234-56-78
GSM: 051/123-456
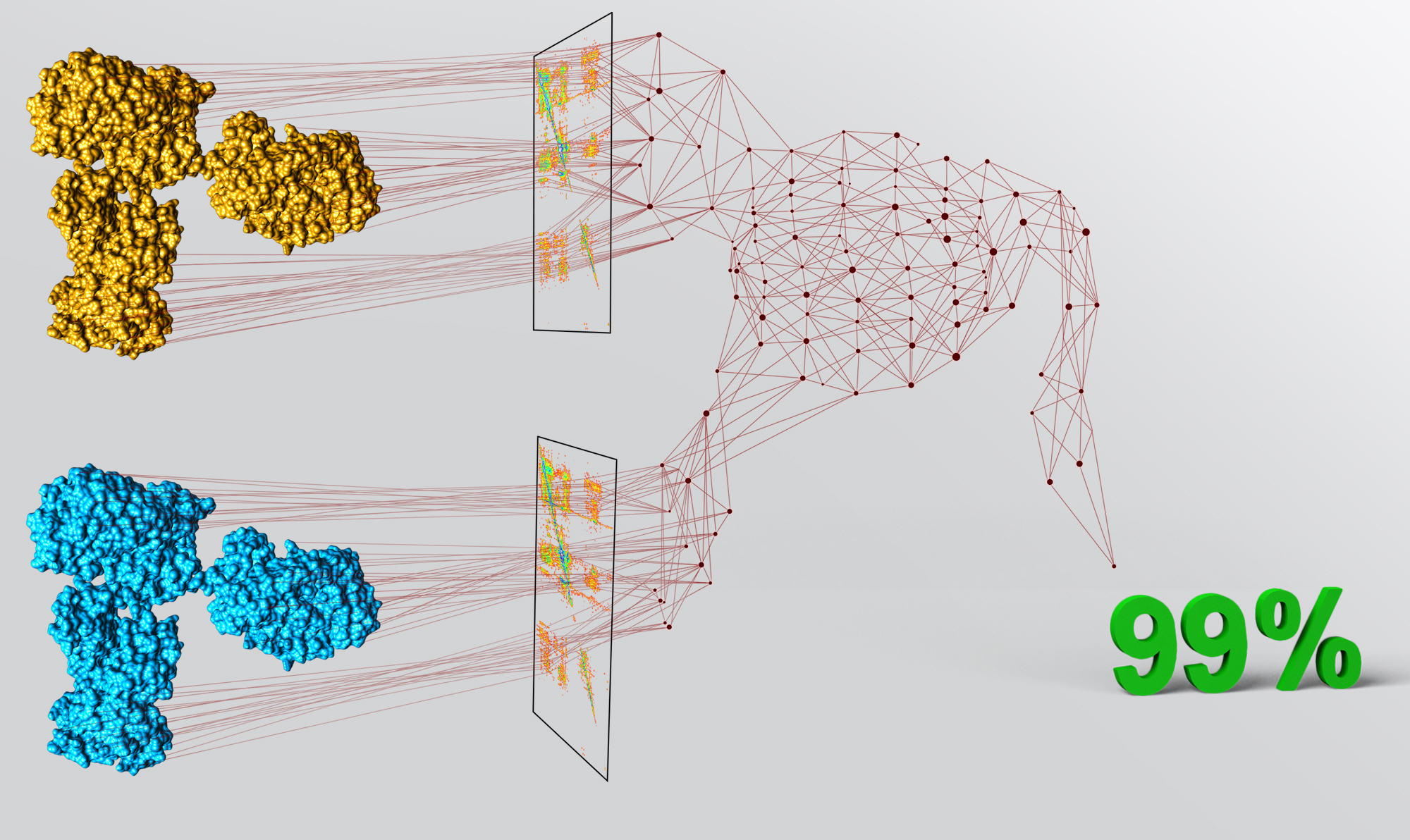
A biosimilar product is a biological product that is approved based on high similarity to an FDA-approved originator product. Biosimilar biological products have to be proven for similarity to the originator molecules in terms of effectiveness and safety for their use in patients. We developed NMR methods that selectively saturate excipients signals in spectra, and thus we are able to use apo form of drug compound in experiments. Combination of highly sensitive NMR fingerprinting methods for conformational changes and bioinformatic techniques allow us to translate complex spectral information into biosimilarity scores that quantify match between biosimilar and originator molecules.